PureSkin
Critical solutions for burn management
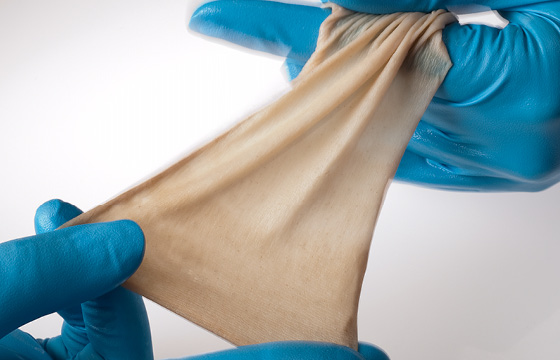
The gold standard in burn care – Our cryopreserved allograft from AlloSource®
AlloSource® has helped save the lives of thousands of patients suffering from traumatic burns and helped restore mobility to countless others. A recognised global leader in the processing and distribution of skin allografts for burn management and wounds resulting from burn injuries, AlloSource® has become one of the largest suppliers of skin allografts used for these applications.
- Minimal prep time – Ready-to-use
- Consistent sizing and thickness – Easy to use and reliable quality
- Flexible meshing options to suit – Non-meshed (meshed available on request)
- Logistics – Same day and flexible options to help manage your time more effectively
For the out-of-hours service please call 01793 575 041.
We are licensed with the Human Tissue Authority (HTA) and recognise the importance of robust quality standards.
AlloSource requires a comprehensive donor physical assessment and a complete medical and social history to identify and eliminate donors that may be at risk of transmitting certain viruses and diseases. Our donor acceptance criteria are based on regulations established by the U.S. Food and Drug Administration (FDA), the American Association of Tissue Banks (AATB) Standards, as well as additional requirements set by the AlloSource Medical Advisory Board.
Donors must test negative or non-reactive in the following assays:
- Antibody to Hepatitis C (HCV)
- Antibody to Human Immunodeficiency Virus 1 & 2 (HIV 1 & 2)
- Hepatitis B Core IgG/IgM Antibody (HBcAb)
- Hepatitis B Surface Antigen (HBsAg)
- Hepatitis C Virus (HCV NAT)
- Human Immunodeficiency Virus Type 1 (HIV-1 NAT)
- Rapid Plasma Reagin or Serologic Test for Syphilis (RPR or STS)
For the out-of-hours service please call 01793 575 041.